Diabetes and Visual Dysfunction (OTHER)

|
John D. Tassinari, OD, FAAO, FCOVD, Dip (BVPPO) AAO
Dr. Tassinari is a lifetime Californian who grew up in the San Fernando Valley. His Bachelor of Science Degree is from Cal State Univ Northridge and his doctor of optometry degree is from the So. Calif College of Optometry. Upon completion of his doctoral program he completed a 1-year residency in Vision Therapy at SUNY College of Optometry in Manhattan. Dr. Tassinari operated a private optometry practice in Pasadena CA from 1988 to 2015. His practice specialized in Vision Therapy and pediatric optometry. During that same time period, Dr. Tassinari served as a part time faculty member for the So Calif College of Optometry. He rose to the rank of assoc. professor in 2013. In 2016, Dr. Tassinari joined the Western University of Health Sciences College of Optometry full time and serves as the chief of pediatrics in the Eye Care Institute. One of his career highlights was attaining the status of Diplomate in the Binocular Vision Perception and Pediatric Optometry Section of the American Academy of Optometry. |
For a printable copy of this article, click here.
Diabetes and Visual Dysfunction
Diabetes is a common metabolic disease which can cause notable deleterious effects on the eyes and vision. Diabetic retinopathy tops the list of diabetic eye diseases that grabs the attention and clinical acumen of optometrists. Diabetes also causes disorders of refraction, accommodation, and binocular vision which will be the focus of this article.
CASE REPORT, PART I
AD, age 32 male, presented to my office wanting to know if his visual symptoms could be resolved with vision therapy (VT). His chief complaint was visual discomfort and fatigue while reading with secondary nearpoint complaints of blur and double vision. The onset of his nearpoint complaints was gradual over the past six months and coincident with a higher workload in his human resources occupation. Two aspects of his case history were a bit confusing. First, he was a low spherical myope (1.00D each eye) who functioned at near unaided. In my experience, low myopes are typically symptom-free for near work. Second, he seemed to believe that his nearpoint symptoms and myopia were one in the same and that VT would cure all of it, including the myopia. AD reported good health and no medications. He commented “I could stand to lose 15-20 pounds” and that his last physical was “oh, one to two years ago.” He has single vision glasses, -1.00DS OU, that he wears part time distance only. My records from this first exam do not show that I asked at what age he first obtained glasses (became myopic). Nor did I ask if his distance vision blur fluctuated. Both questions may have led to a quicker connection of the dots. Namely, adult age 30+ onset myopia with fluctuating distance blur is highly suggestive of diabetes.1, 2
My exam showed no change in his spectacle prescription and normal best corrected visual acuities. Color vision, stereoacuity and visual fields were normal. Cover test was orthophoria at distance and near. External exam, biomicroscopy and funducopy through dilated pupils, were unremarkable. Approximately 30 minutes after instilling tropicamide 1 percent and hydroxyamphetamine 1 percent with .25 percent tropicamide (Paremyd); I demonstrated +.25DS monocularly while wearing his glasses and it caused a slight blur. Plus .50DS in the same manner was definitely blurry. This cycloplegic trial frame refraction ruled out pseudomyopia and the possibility of VT reversing the myopia.
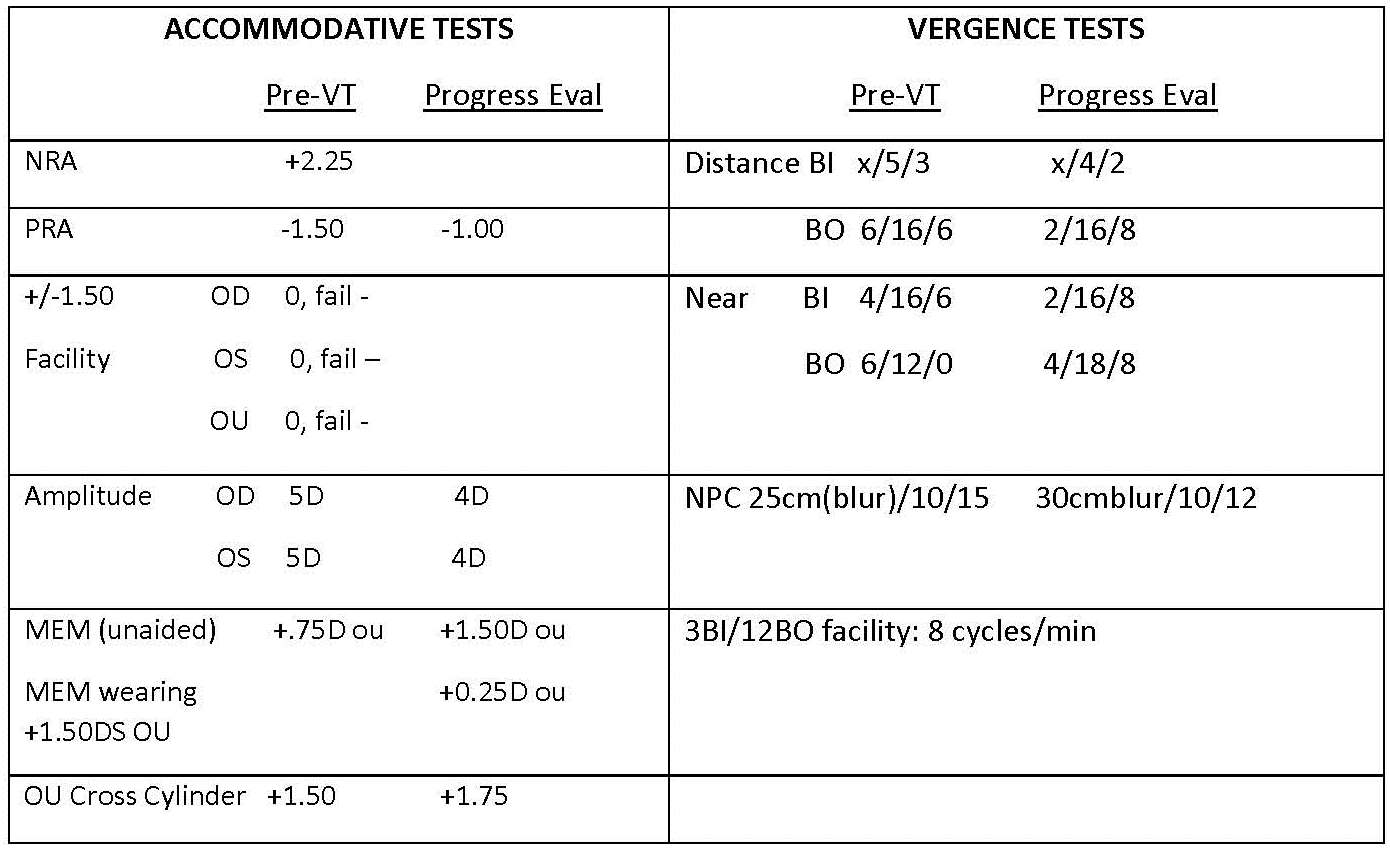
The remarkable findings from my exam were low scores on several tests of accommodation and vergence as shown in Table 1. Note the low blur points on the vergence range tests and his inability to clear the minus side of the accommodative facility test. Accommodative amplitudes, using a modified push-up minus lens method, 3 were very low. One other nuance to note in the exam data is the accommodative lag per monocular estimate method (MEM) nearpoint retinoscopy. While the result, +.75, is not considered abnormally high, it took place unaided. With the target at 40 cm and AD sans his 1.00D OU myopic correction, the accommodative demand was 1.50D. In response to this demand, AD activated (only) .75D of accommodation which explains why a .75 lag was observed. I diagnosed AD with symptomatic accommodative insufficiency and fusional vergence dysfunction. Treatment options are a plus add, VT, or combination of both. He already had an effective plus add because of uncorrected myopia for near vision. A higher add is an option but I was confident VT alone would resolve his symptoms. It took an inordinate amount of time and all my powers of communication to explain that VT would not cure his myopia. Its goal is to improve accommodative and vergence function and ameliorate his nearpoint symptoms. AD assented and enrolled in an office-based VT program.
AD’s attendance to the once-weekly scheduled office VT sessions was sporadic. Two-and-one-half months passed and he had only completed three VT sessions. My vision therapist informed me that AD had an urgent request to move up his progress evaluation because he is noticing significant visual changes. At his third VT office visit he told my therapist, “It’s getting worse and my glasses don’t work.”
At this office visit, AD was quite distressed. “My glasses make everything blurry. I stopped using them even though you told me I would always need them.” Upon further questioning, he was particularly upset because they made near vision blurry. Confused, I reminded him that they are not for near vision. He replied, “At least before, I could look at my watch or my phone with my glasses on. Now I can’t.” He further complained that his unaided near vision was much worse and his eyes tire quickly with reading “way worse than before I began VT.” He wanted to know if VT made his near vision worse. I was further confused when I saw that my assistant had tested his unaided distance vision and it showed 20/20 monocular visual acuity and 20/15 binocular. I began testing him and found a distance refraction of +.50DS each eye. I continued my exam with distance vergence ranges following the refraction. Pondering the low blur points (Table 2) and proceeding to attach the nearpoint rod suddenly the proverbial light bulb glowed above my head. I pulled the phoropter away and asked him if there were changes in his medical status. “I have diabetes,” he replied, “not like actual diabetes but pre-diabetes.” He further explained that he has been taking metformin 500mg BID for two weeks. It was in that moment that I clarified that he obtained his distance glasses nine months before my first exam and that his weight gain began about one year ago. I further questioned him about fluctuating distance vision and he answered “Well, there were times when the glasses felt too strong.” I continued my exam and as shown in Table 1, his accommodative and vergence function were worse on some measures.
Table 1. Vision Therapy (VT) evaluation tests.
REFRACTIVE CHANGES AND DIABETES
AD had a classic transient myopic shift secondary to diabetes-caused hyperglycemia. It resolved when his circulating blood glucose returned to normal levels with medication. The myopic shift that takes place in diabetes is due to changes within the crystalline lens. It’s not necessarily that the crystalline lens is damaged like other organs and tissues in diabetes. It’s that the extra circulating glucose, present because insulin-resistant cells are not ingesting it, alters the sorbitol and dulcitol pathways of the crystalline lens, which in turn swells and/or changes the refractive index of the lens nucleus.1, 2 Once homeostasis is restored to the dysregulated circulating glucose, the normal biochemistry and dioptric power of the crystalline lens is restored which cures the transitory myopia. Of note, hyperopic shifts from hyperglycemia have been reported.4 This type of refractive change is thought to be due to the same biochemical processes occurring in one or more of the layers of the crystalline lens cortex.
ACCOMMODATION/VERGENCE AND DIABETES
In my opinion, AD’s accommodation and vergence dysfunction were also sequelae of his diabetes. In support of this cause and effect is the coincidence of his visual discomfort/fatigue symptoms with the myopia and presumed onset of hyperglycemia. Indeed, their coincidental onset led to AD correctly concluding they were one in the same visual problem. There is plenty of literature support for accommodative dysfunction and diabetes.2,3,5,6 One study went so far as to measure accommodative amplitude with visually evoked cortical potential as opposed to subjective report of blur.6 It showed that diabetics have a statistically significant reduced amplitude of accommodation compared to age matched controls (p < .01). Another strength of this study is that the adult diabetic subjects were all 20/20 best corrected and had no diabetic retinopathy (n=10) or no worse than mild non-proliferative diabetic retinopathy (n=22). The locus of diabetic accommodative dysfunction is not known. Because it is bilateral and equal between the two eyes, and there are no other third nerve signs, e.g., ptosis or exotropia, one might speculate that the Edinger-Westphal nucleus within the third nerve nuclear complex is selectively lesioned. Further evidence for a lesion to Edinger-Westphal nucleus would be compromised parasympathetic efferent signal to pupillary constrictor smooth muscles. Clinically, this compromise is seen as bilateral sluggish pupil reaction to light during the swinging flashlight test.7,8
Fusional vergence dysfunction, low ranges in the presence of a normal heterophoria, is a lesser known and lesser understood visual sequelae of diabetes.3 It could simply be that the intimate link between accommodation and vergence means that accommodative dysfunction is primary and it drags down vergence function along with it. Keep in mind that acquired vergence dysfunction, e.g., a decline in vergence skills due to illness, is not the same as acquiring a strabismus due to a disease process. Diabetes is known to cause neuroparalytic strabismus with pupil-sparing third nerve and sixth nerve paresis being the most and roughly equal types.9 Diabetes may also cause gaze evoked strabismus if there is infarct to the medial longitudinal fasiculus.9 This disease process is called internuclear ophthalmoplegia.
If hyperglycemia myopia resolves with successful medication, does diabetes induce acquired accommodative and vergence dysfunction? The best answer is “maybe not.” Patients with controlled diabetes are prone to accommodative dysfunction as the study above points out.6 Sluggish iris constriction is not noticed to improve with controlled diabetes. The case in this article provides a definitive answer of “no,” at least in the short term, as described below.
CASE REPORT PART II
While AD’s transient myopia dissipated with diabetes medication, his accommodative and vergence dysfunction did not. Table II shows that his accommodative and vergence function are worse on some measures. Furthermore, his symptoms increased because he lost the myopia near vision advantage. He was anxious and eager to gain symptomatic relief so I proceeded to determine a nearpoint lens. His eagerness aside, it is also true that accommodative dysfunction due to an underlying illness prompts me to lean toward a plus add as the sole treatment or in conjunction with VT. Using a blend of the binocular cross cylinder, MEM retinoscopy, and the subjective refraction result of .50D hyperopia, I prescribed +1.50D spheres OU. AD pre-emptively declined continuation of VT before I could recommend it and did not seem to have complete confidence in my explanation of his diabetes and visual problems. He interrupted me a few times to correct my usage of the term diabetes. He did not have diabetes, he had pre-diabetes, which seemed to weaken his belief in my conclusions. He established himself with a retina specialist ophthalmologist who requested my records and I did not have a chance to re-evaluate him. It would be interesting to learn if his accommodative and vergence dysfunction improved over time.
CONCLUSION
Diabetes is a metabolic disorder in which cells within any organ can be starved of proper nutrition because glucose cannot pass through the cell membrane due to lack of the hormone insulin (Type 1) or cells inexplicably become insulin resistant (Type 2). The cells of eye and its complex neural network may be one of those organs and in certain cases visual function may be problematic while no structural eye abnormalities are observed. Transient myopia can simply be waited out. A reasonable approach to accommodative dysfunction is to prescribe a plus add and if symptoms are not relieved, consider VT as an adjunct treatment for accommodative dysfunction.
REFERENCES
1. Maija Mäntyjärvi M. Myopia and diabetes: A review. Acta Ophthalmologica. 1988; 66: 82-85.
2. Cavallerano JD. A review of non-retinal ocular complications of diabetes mellitus. J Amer Optom Assoc. 1991; 61: 533-43
3. Scheiman S, Wick B. Clinical management of binocular vision: heterophoric, accommodative, and eye movement disorders. Philadelphia, Lippincott Williams & Wilkins. 2008
4. Fledelius HC. Refractive change in hyperglycaemia: hyperopia not myopia. Brit J Ophthalmol. 1982; 66: 500-505.
5. Marmor MF. Transient Accommodative Paralysis and Hyperopia in Diabetes. Arch Ophthalmol. 1973 ;89(5):419-421.
6. Yamamoto S, Adachi-Usami E, Kuroda N. Accommodation power determined with transient pattern visual evoked cortical potentials in diabetes. Documenta Opthalmologica. 1989; 72 (1) 31–37.
7. Spafford MM, Lovasik JV. Clinical evaluation of ocular and visual functions in insulin-dependent juvenile diabetics. Amer J Optom Physio Optics. 1986; 63: 505-19.
8. Wilhelm, H. Neuro-ophthalmology of pupillary function-practical guidelines. J Neurol. 1998; 245: 573-583.
9. Friedman, SA et al. Pupillary Abnormalities in Diabetic Neuropathy: A Preliminary Study. Ann Intern Med. 1967; 67 (5):977-983.
10. Miller NR, Newman NJ. Walsh & Hoyt’s Clinical Neuro-Ophthalmology 5th ed. Baltimore, Williams & Wilkins. 1999
11. Okamato F, Sone H, et. al. Refractive changes in diabetic patients during intensive glycaemic control. Br J Ophthalmol. 2000; 84: 1097-1102.
12. Lin SF, Lin PK, et.al. Transient hyperopia after intensive treatment of hyperglycemia in newly diagnosed diabetes. Ophthalmologica. 2009; 223: 68-71.
| Click HERE to take the test |
1.png)

1.png)



.png)




.png)
.png)
.png)
.jpg)
.png)

.png)
.png)
.png)
.png)
.png)

.png)

.png)
.png)
